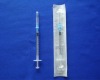
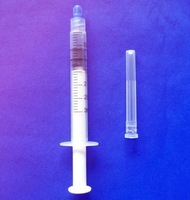
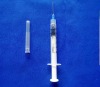
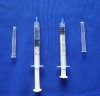
plastic medical disposable syringe 1ml: WHO,CE,ISO remark With needle, protective cap Clear scale on barrel easy to read
Detailed information: Plastic medical disposable syringe 1ml / AD syringe
(with CE, WHO (PQC) and ISO13485 certificate)
Specification:
0.05ml, 0.3ml, 0.5ml, 1ml
It is a self-destruction syringe .Price is as competitive as normal syringe. It can fully use the liquid nearly 100%. It can prevent the crossing infection .After injection, the destruction part in the barrel will pierce the plunger, and then when you try to pump the liquid again, it is leaking and can not use again.
Package: PE and Blister
Valid for 3 years only.
1.Graduated scale on the barrel easy to read.
2.with needle and protective cap
3.Material:
Syringe: Polypropylene (pp)
Needle: Stainless steel, Disposable
Sterile (ethylene oxide sterilization)
Packaging and labeling
Primary packaging: Unit of use
1 sterile syringe packed in an individual sterilized peel-pack made of Blister paper and film, 10 sterile blister and PE pack syringe attached with each other but each syringe can be separated easily.
Labeling on the primary packaging:
1.Name and/or trade mark of the manufacturer.
2.The word "sterile"
3.Sterilization method
4.Expiry date by year and month, prefixed by the word "EXP"
5.The words "for single use"
6.The words "check the integrity of the individual sterilized pack before use"
Instructions for use:
1 Injection safety:
2.The syringe is sterile, ready for immediate use and is for single use ONLY
3.Check the integrity of the packaging before opening.
4.Do not use the syringe if the packaging is not sealed or is pierced.
plastic medical disposable syringe 1ml